BU-106a: Choices of Primary Batteries

Zinc-carbon, also known as carbon-zinc or the Leclanché battery, is one of the earliest and least expensive primary batteries. It delivers 1.5V and often come with consumer devices. The first zinc-carbon invented by Georges Leclanché in 1859 was wet.
Alkaline. Alkaline-manganese, also known as alkaline, is an improved version of the zinc-carbon battery and delivers 1.5V. Lewis Urry (1927–2004) invented alkaline in 1949 while working with the Eveready Battery Company laboratory in, Ohio, USA.
Alkaline delivers more energy at higher load currents than zinc-carbon. Furthermore, a regular household alkaline provides about 40 percent more energy than the average Li-ion but alkaline is not as strong as Li-ion on loading. Alkaline has very low self-discharge and does not leak electrolyte when depleted as the old zinc-carbon does, but it is not totally leak-proof.
All primary batteries produce a small amount of hydrogen gas on discharge and battery-powered devices must make provision for venting. Pressure buildup in the cell can rupture the seal and cause corrosion. This is visible in form of a feathery crystalline structure that can develop and spread to neighboring parts in the device and cause damage.
Lithium iron disulfide (Li-FeS2) is a newcomer to the primary battery family and offers improved performance compared to alkaline. Lithium batteries normally deliver 3 volts and higher, but Li-FeS2 has 1.5 volts to be compatible with the AA and AAA formats. It has a higher capacity and a lower internal resistance than alkaline. This enables moderate to heavy loads and is ideal for digital cameras. Further advantages are improved low temperature performance, superior leakage resistance and low self-discharge, allowing 15 years of storage at ambient temperatures.
The disadvantages of the Li-FeS2 are a higher price and transportation issues due to the lithium metal content in the anode. In 2004, the US DOT and the Federal Aviation Administration (FAA) banned bulk shipments of primary lithium batteries on passenger flights, but airline passengers can still carry them on board if the allotted lithium content is not exceeded. Each AA-sized Li-FeS2 contains 0.98 grams of lithium; the air limitation of primary lithium batteries is 2 grams (8 grams for rechargeable Li-ion). This restricts each passenger to two cells, but exceptions have been made in which 12 sample batteries can be carried. (See BU-704a: Shipping Lithium-based Batteries by air)
The Li-FeS2 includes safety devices in the form of a positive thermal coefficient (PTC) that limits the current at high temperature and resets when normal. The Li-FeS2 cell cannot be recharged as is possible with NiMH in the AA and AAA formats. Recharging, putting a cell in backwards, mixing in a depleted cell or adding a foreign cell could cause a leak or explosion. (See BU-304a: Safety Concerns with Li-ion)
Figures 1 and 2 compare the discharge voltage and internal resistance of alkaline and Li-FeS2 at a 50mA pulsed load. Of interest is the flat voltage curve and the low internal resistance of lithium; alkaline shows a rapid voltage drop and a permanent increase in resistance with use. This shortens the runtime, especially at an elevated load.
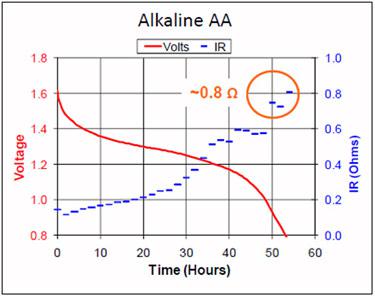
Figure 1: Voltage and internal resistance of alkaline on discharge.
The internal resistance rises, causing the voltage to drop. Source: Energizer
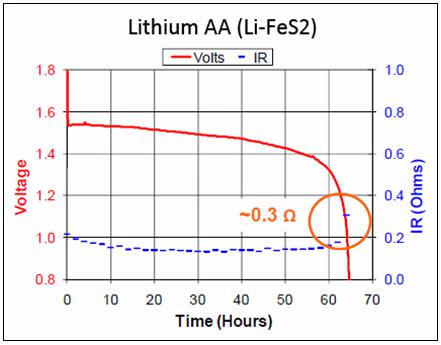
Figure 2: Voltage and internal resistance of lithium on discharge.
The internal resistance remains low and the voltage stays flat. Source: Energizer
Lithium-thionyl chloride (LiSOCI2 or LTC) is one of the most rugged lithium-metal batteries. The ability to withstand high heat and strong vibration enables horizontal drilling, also known as fracking. Some LTC are said to operate from 0°C to 200°C (32°F to 392°F). Other uses are in medical and sensor applications.
With a specific energy of over 500Wh/kg, LTC offers twice the capacity of the best Li-ion. The nominal voltage is 3.60V/cell; the end-of-discharge cut-off voltage is 3.00V. The runtime is not based on capacity alone; thermal conditions and load pattern also have an effect. Constant current is more enduring than pulsed load; a phenomenon that applies to most batteries.
Like alkaline, lithium-thionyl chloride has a relatively high resistance and can only be used for moderate discharge loads. If stored for a time, a passivation layer forms between the lithium anode and the carbon-based cathode that dissipates when applying a load. This layer protects the battery by granting low self-discharge and a long shelf life. (See BU-701: How to Prime Batteries)
LTC is one of the most powerful and potent battery chemistries and should only be used by trained workers. For safety reasons, this battery is not used in consumer devices.
Lithium manganese dioxide (LiMnO2 or Li-M) is similar to LTC but has a lower specific capacity and is safe for public use. The voltage is 3.0–3.30V and the specific energy is about 280Wh/kg. Li-M is economically priced, has a long life and allows moderate loads but can deliver high pulse currents. Operational temperature ranges from -30°C to 60°C (-22°F to 140°F). Typical uses are meter sensing, medical devices, road toll sensors and cameras.
Lithium sulfur dioxide (LiSo2) is a primary battery with a voltage of 2.8V and an energy density up to 330Wh/kg. It offers a wide temperature range of --54°C to 71°C (-65°F to 160°F) with a projected shelf life of 5–10 years at room temperature. LiSo2 is inexpensive to make and is commonly used by the military. The Iraqi war used tons of these batteries, but it is giving way to the more superior Li-M.
Note: Primary lithium batteries are also known as lithium-metal. The cathode is carbon and the anode holds the active material, the reverse of Li-ion, which features a carbon anode.
Table 3 summarizes the most common primary batteries.
| Primary Cell | Alkaline | Lithium iron disulfide (LiFeS2) | Lithium-thionyl chloride (LiSOCI2 or LTC) | Lithium manganese dioxide (LiMnO2 or Li-M) | Lithium sulfur dioxide |
|---|---|---|---|---|---|
| Specific energy | 200Wh/kg | 300Wh/kg | 500Wh/kg | 280Wh/kg | 330Wh/kg |
| Voltage | 1.5V | 1.5V | 3.6–3.9V | 3–3.3V | 2.8V |
| Power | Low | Moderate | Excellent | Moderate | Moderate |
| Passivation | N/A | Moderate | Moderate | Moderate | Moderate |
| Safety | Good | Good | Precaution | Good | Precaution |
| Pricing | Low | Economical | Industrial | Economical | Industrial |
| Shelf life | 10 years | 15 years | 10–20 years | 10–20 years | 5–10 years |
| Operating temp | 0°C to 60°C | 0°C to 60°C | -55°C to 85°C, higher for short time | -30°C to 60°C some enable from -55°C to 90°C | -54°C to 71°C |
| Usage | Consumer devices | Swaps alkaline for higher power and long runtime | Horizontal drilling, (fracking). Not for consumer use. | Meter sensing, medical devicaes, road toll sensors, cameras | Defense; being replaced by LiMnO2 |
Table 3: Summary table of common primary batteries. Values are estimated.
LTC and Li-M are safe but workers handling these batteries must be familiar with safety precautions, transportation and disposal. Protect the batteries from heat, short circuit, and physical or electrical abuses.
Last Updated: 21-Oct-2021

Batteries In A Portable World
The material on Battery University is based on the indispensable new 4th edition of "Batteries in a Portable World - A Handbook on Rechargeable Batteries for Non-Engineers" which is available for order through Amazon.com.
Find An Article
Table of Contents
-
Introduction
- BU-001: Sharing Battery Knowledge
- BU-002: Introduction
- BU-003: Dedication
-
Crash Course on Batteries
- BU-101: When Was the Battery Invented?
- BU-102: Early Innovators
- BU-103: Global Battery Markets
- BU-103a: Battery Breakthroughs: Myth or Fact?
- BU-104: Getting to Know the Battery
- BU-104a: Comparing the Battery with Other Power Sources
- BU-104b: Battery Building Blocks
- BU-104c: The Octagon Battery – What makes a Battery a Battery
- BU-105: Battery Definitions and what they mean
- BU-106: Advantages of Primary Batteries
- BU-106a: Choices of Primary Batteries
- BU-107: Comparison Table of Secondary Batteries
-
Battery Types
- BU-201: How does the Lead Acid Battery Work?
- BU-201a: Absorbent Glass Mat (AGM)
- BU-201b: Gel Lead Acid Battery
- BU-202: New Lead Acid Systems
- BU-203: Nickel-based Batteries
- BU-204: How do Lithium Batteries Work?
- BU-205: Types of Lithium-ion
- BU-206: Lithium-polymer: Substance or Hype?
- BU-208: Cycling Performance
- BU-209: How does a Supercapacitor Work?
- BU-210: How does the Fuel Cell Work?
- BU-210a: Why does Sodium-sulfur need to be heated
- BU-210b: How does the Flow Battery Work?
- BU-211: Alternate Battery Systems
- BU-212: Future Batteries
- BU-214: Summary Table of Lead-based Batteries
- BU-215: Summary Table of Nickel-based Batteries
- BU-216: Summary Table of Lithium-based Batteries
- BU-217: Summary Table of Alternate Batteries
- BU-218: Summary Table of Future Batteries
-
Packaging and Safety
- BU-301: A look at Old and New Battery Packaging
- BU-301a: Types of Battery Cells
- BU-302: Series and Parallel Battery Configurations
- BU-303: Confusion with Voltages
- BU-304: Why are Protection Circuits Needed?
- BU-304a: Safety Concerns with Li-ion
- BU-304b: Making Lithium-ion Safe
- BU-304c: Battery Safety in Public
- BU-305: Building a Lithium-ion Pack
- BU-306: What is the Function of the Separator?
- BU-307: How does Electrolyte Work?
- BU-308: Availability of Lithium
- BU-309: How does Graphite Work in Li-ion?
- BU-310: How does Cobalt Work in Li-ion?
- BU-311: Battery Raw Materials
-
Charge Methods
- BU-401: How do Battery Chargers Work?
- BU-401a: Fast and Ultra-fast Chargers
- BU-402: What Is C-rate?
- BU-403: Charging Lead Acid
- BU-404: What is Equalizing Charge?
- BU-405: Charging with a Power Supply
- BU-406: Battery as a Buffer
- BU-407: Charging Nickel-cadmium
- BU-408: Charging Nickel-metal-hydride
- BU-409: Charging Lithium-ion
- BU-409a: Why do Old Li-ion Batteries Take Long to Charge?
- BU-409b: Charging Lithium Iron Phosphate
- BU-410: Charging at High and Low Temperatures
- BU-411: Charging from a USB Port
- BU-412: Charging without Wires
- BU-413: Charging with Solar, Turbine
- BU-413a: How to Store Renewable Energy in a Battery
- BU-414: How do Charger Chips Work?
- BU-415: How to Charge and When to Charge?
-
Discharge Methods
- BU-501: Basics about Discharging
- BU-501a: Discharge Characteristics of Li-ion
- BU-502: Discharging at High and Low Temperatures
- BU-503: Determining Power Deliver by the Ragone Plot
- BU-504: How to Verify Sufficient Battery Capacity
-
"Smart" Battery
- BU-601: How does a Smart Battery Work?
- BU-602: How does a Battery Fuel Gauge Work?
- BU-603: How to Calibrate a “Smart” Battery
- BU-603a: Calibrating SMBus Batteries with Impedance Tracking
- BU-604: How to Process Data from a “Smart” Battery
- BU-605: Testing and Calibrating Smart Batteries
-
From Birth to Retirement
- BU-701: How to Prime Batteries
- BU-702: How to Store Batteries
- BU-703: Health Concerns with Batteries
- BU-704: How to Transport Batteries
- BU-704a: Shipping Lithium-based Batteries by Air
- BU-704b: CAUTION & Overpack Labels
- BU-704c: Class 9 Label
- BU-704d: NFPA 704 Rating
- BU-704e: Battery for Personal and Fleet Use
- BU-705: How to Recycle Batteries
- BU-705a: Battery Recycling as a Business
- BU-706: Summary of Do's and Don'ts
-
How To Prolong Battery Life
-
General
- BU-801: Setting Battery Performance Standards
- BU-801a: How to Rate Battery Runtime
- BU-801b: How to Define Battery Life
- BU-802: What Causes Capacity Loss?
- BU-802a: How does Rising Internal Resistance affect Performance?
- BU-802b: What does Elevated Self-discharge Do?
- BU-802c: How Low can a Battery be Discharged?
- BU-803: Can Batteries Be Restored?
- BU-803a: Cell Matching and Balancing
- BU-803b: What causes Cells to Short?
- BU-803c: Loss of Electrolyte
-
Lead Acid
- BU-804: How to Prolong Lead-acid Batteries
- BU-804a: Corrosion, Shedding and Internal Short
- BU-804b: Sulfation and How to Prevent it
- BU-804c: Acid Stratification and Surface Charge
- BU-805: Additives to Boost Flooded Lead Acid
- BU-806: Tracking Battery Capacity and Resistance as part of Aging
- BU-806a: How Heat and Loading affect Battery Life
-
Nickel-based
- BU-807: How to Restore Nickel-based Batteries
- BU-807a: Effect of Zapping
-
Lithium-ion
- BU-808: How to Prolong Lithium-based Batteries
- BU-808a: How to Awaken a Sleeping Li-ion
- BU-808b: What Causes Li-ion to Die?
- BU-808c: Coulombic and Energy Efficiency with the Battery
- BU-809: How to Maximize Runtime
- BU-810: What Everyone Should Know About Aftermarket Batteries
- BU-811: Assuring Minimum Operational Reserve Energy (MORE)
-
Battery Testing and Monitoring
- BU-901: Fundamentals in Battery Testing
- BU-901b: How to Measure the Remaining Useful Life of a Battery
- BU-902: How to Measure Internal Resistance
- BU-902a: How to Measure CCA
- BU-903: How to Measure State-of-charge
- BU-904: How to Measure Capacity
- BU-905: Testing Lead Acid Batteries
- BU-905a: Testing Starter Batteries in Vehicles
- BU-905b: Knowing when to Replace a Starter Battery
- BU-906: Testing Nickel-based Batteries
- BU-907: Testing Lithium-based Batteries
- BU-907a: Battery Rapid-test Methods
- BU-907b: Advancements in Battery Testing
- BU-907c: Cloud Analytics in Batteries
- BU-908: Battery Management System (BMS)
- BU-909: Battery Test Equipment
- BU-910: How to Repair a Battery Pack
- BU-911: How to Repair a Laptop Battery
- BU-915: Testing Battery with EIS
- BU-916: Deep Battery Diagnostics
- BU-917: In Search for Performance Transparency with Batteries
- BU-918: Battery Endurance Plan
- BU-919: Building a Matrix to test Batteries
- BU-920: Matrix Library
- BU-921: Testing Batteries by Multi-Model EIS
- BU-922: What Causes Starter Batteries to Fail?
- BU-923: Getting Deep Cycle Batteries Mission Ready
- BU-924: Battery Ecosystem
- BU-925: EIS Direct Drive, non-invasive Capacity Test
- BU-926: Battery Diagnostics, an Overlooked Requirement
-
Amazing Value of a Battery
- BU-1001: Batteries in Industries
- BU-1002: Electric Powertrain, then and now
- BU-1002a: Hybrid Electric Vehicles and the Battery
- BU-1002b: Environmental Benefit of the Electric Powertrain
- BU-1003: Electric Vehicle (EV)
- BU-1003a: Battery Aging in an Electric Vehicle (EV)
- BU-1004: Charging an Electric Vehicle
- BU-1005: Does the Fuel Cell-powered Vehicle have a Future?
- BU-1006: Cost of Mobile and Renewable Power
- BU-1007: Net Calorific Value
- BU-1008: Working towards Sustainability
- BU-1009: Battery Paradox - Afterword
-
Information
- BU-1101: Glossary
- BU-1102: Abbreviations
- BU-1103: Bibliography
- BU-1104: About the Author
- BU-1105: About Cadex (Sponsor)
- BU-1106: Author's Creed
- BU-1107: Disclaimer
- BU-1108: Copyright
-
Learning Tools
- BU-1501 Battery History
- BU-1502 Basics about Batteries
- BU-1504 Battery Test and Analyzing Devices
- BU-1505 Short History of Cadex
-
Battery Articles
- Perception of a Battery Tester
- Green Deal
- Risk Management in Batteries
- Predictive Test Methods for Starter Batteries
- Why Mobile Phone Batteries do not last as long as an EV Battery
- Battery Rapid-test Methods
- How to Charge Li-ion with a Parasitic Load
- Ultra-fast Charging
- Assuring Safety of Lithium-ion in the Workforce
- Diagnostic Battery Management
- Tweaking the Mobile Phone Battery
- Battery Test Methods
- Battery Testing and Safety
- How to Make Battery Performance Transparent
- Battery Diagnostics On-the-fly
- Making Battery State-of-health Transparent
- Batteries will eventually die, but when and how?
- Why does Pokémon Go rob so much Battery Power?
- How to Care for the Battery
- Tesla’s iPhone Moment — How the Powerwall will Change Global Energy Use
- Painting the Battery Green by giving it a Second Life
- Charging without Wires — A Solution or Laziness
- What everyone should know about Battery Chargers
- A Look at Cell Formats and how to Build a good Battery
- Battery Breakthroughs — Myth or Fact?
- Rapid-test Methods that No Longer Work
- Shipping Lithium-based Batteries by Air
- How to make Batteries more Reliable and Longer Lasting
- What causes Lithium-ion to die?
- Safety of Lithium-ion Batteries
- Recognizing Battery Capacity as the Missing Link
- Managing Batteries for Warehouse Logistics
- Caring for your Starter Battery
- Giving Batteries a Second Life
- How to Make Batteries in Medical Devices More Reliable
- Possible Solutions for the Battery Problem on the Boeing 787
- Impedance Spectroscopy Checks Battery Capacity in 15 Seconds
- How to Improve the Battery Fuel Gauge
- Examining Loading Characteristics on Primary and Secondary Batteries
-
Language Pool
- BU-001: Compartir conocimiento sobre baterías
- BU-002: Introducción
- BU-003: Dedicatoria
- BU-104: Conociendo la Batería
- BU-302: Configuraciones de Baterías en Serie y Paralelo
-
Batteries in a Portable World book
- Change-log of “Batteries in a Portable World,” 4th edition: Chapters 1 - 3
- Change-log of “Batteries in a Portable World,” 4th edition: Chapters 4 - 10