BU-808: How to Prolong Lithium-based Batteries

Battery research is focusing on lithium chemistries so much that one could imagine that the battery future lies solely in lithium. There are good reasons to be optimistic as lithium-ion is, in many ways, superior to other chemistries. Applications are growing and are encroaching into markets that previously were solidly held by lead acid, such as standby and load leveling. Many satellites are also powered by Li-ion.
Lithium-ion has not yet fully matured and is still improving. Notable advancements have been made in longevity and safety while the capacity is increasing incrementally. Today, Li-ion meets the expectations of most consumer devices but applications for the EV need further development before this power source will become the accepted norm. BU-104c: The Octagon Battery – What makes a Battery a Battery, describes the stringent requirements a battery must meet.
As battery care-giver, you have choices in how to prolong battery life. Each battery system has unique needs in terms of charging speed, depth of discharge, loading and exposure to adverse temperature. Check what causes capacity loss, how does rising internal resistance affect performance, what does elevated self-discharge do and how low can a battery be discharged? You may also be interested in the fundamentals of battery testing.
What Causes Lithium-ion to Age?
The lithium-ion battery works on ion movement between the positive and negative electrodes. In theory such a mechanism should work forever, but cycling, elevated temperature and aging decrease the performance over time. Manufacturers take a conservative approach and specify the life of Li-ion in most consumer products as being between 300 and 500 discharge/charge cycles.
In 2020, small wearable batteries deliver about 300 cycles whereas modern smartphones have a cycle life requirement is 800 cycles and more. The largest advancements are made in EV batteries with talk about the one-million-mile battery representing 5,000 cycles.
Evaluating battery life on counting cycles is not conclusive because a discharge may vary in depth and there are no clearly defined standards of what constitutes a cycle(See BU-501: Basics About Discharging). In lieu of cycle count, some device manufacturers suggest battery replacement on a date stamp, but this method does not take usage into account. A battery may fail within the allotted time due to heavy use or unfavorable temperature conditions; however, most packs last considerably longer than what the stamp indicates.
The performance of a battery is measured in capacity, a leading health indicator. Internal resistance and self-discharge also play roles, but these are less significant in predicting the end of battery life with modern Li-ion.
Figure 1 illustrates the capacity drop of 11 Li-polymer batteries that have been cycled at a Cadex laboratory. The 1,500mAh pouch cells for mobile phones were first charged at a current of 1,500mA (1C) to 4.20V/cell and then allowed to saturate to 0.05C (75mA) as part of the full charge saturation. The batteries were then discharged at 1,500mA to 3.0V/cell, and the cycle was repeated. The expected capacity loss of Li-ion batteries was uniform over the delivered 250 cycles and the batteries performed as expected.

Figure 1: Capacity drop as part of cycling [1]
Eleven new Li-ion were tested on a Cadex C7400 battery analyzer. All packs started at a capacity of 88–94% and decreased to 73–84% after 250 full discharge cycles. The 1500mAh pouch packs are used in mobile phones.
Although a battery should deliver 100 percent capacity during the first year of service, it is common to see lower than specified capacities, and shelf life may contribute to this loss. In addition, manufacturers tend to overrate their batteries, knowing that very few users will do spot-checks and complain if low. Not having to match single cells in mobile phones and tablets, as is required in multi-cell packs, opens the floodgates for a much broader performance acceptance. Cells with lower capacities may slip through cracks without the consumer knowing.
Similar to a mechanical device that wears out faster with heavy use, the depth of discharge (DoD) determines the cycle count of the battery. The smaller the discharge (low DoD), the longer the battery will last. If at all possible, avoid full discharges and charge the battery more often between uses. Partial discharge on Li-ion is fine. There is no memory and the battery does not need periodic full discharge cycles to prolong life. The exception may be a periodic calibration of the fuel gauge on a smart battery or intelligent device(See BU-603: How to Calibrate a “Smart” Battery)
The following tables indicate stress related capacity losses on cobalt-based lithium-ion. The voltages of lithium iron phosphate and lithium titanate are lower and do not apply to the voltage references given.
Tables 2, 3 and 4 indicate general aging trends of common cobalt-based Li-ion batteries on depth-of-discharge, temperature and charge levels, Table 6 further looks at capacity loss when operating within given and discharge bandwidths. The tables do not address ultra-fast charging and high load discharges that will shorten battery life. No all batteries behave the same.
Table 2 estimates the number of discharge/charge cycles Li-ion can deliver at various DoD levels before the battery capacity drops to 70 percent. DoD constitutes a full charge followed by a discharge to the indicated state-of-charge (SoC) level in the table.
Depth of Discharge |
Discharge cycles |
|
|---|---|---|
NMC |
LiPO4 |
|
100% DoD |
~300 |
~600 |
80% DoD |
~400 |
~900 |
60% DoD |
~600 |
~1,500 |
40% DoD |
~1,000 |
~3,000 |
20% DoD |
~2,000 |
~9,000 |
10% DoD |
~6,000 |
~15,000 |
Table 2: Cycle life as a function ofdepth of discharge*
A partial discharge reduces stress and prolongs battery life, so does a partial charge. Elevated temperature and high currents also affect cycle life.
Lithium-ion suffers from stress when exposed to heat, so does keeping a cell at a high charge voltage. A battery dwelling above 30°C (86°F) is considered elevated temperature and for most Li-ion a voltage above 4.10V/cell is deemed as high voltage. Exposing the battery to high temperature and dwelling in a full state-of-charge for an extended time can be more stressful than cycling. Table 3 demonstrates capacity loss as a function of temperature and SoC.
| Temperature | 40% Charge | 100% Charge |
|---|---|---|
| 0°C | 98% (after 1 year) | 94% (after 1 year) |
| 25°C | 96% (after 1 year) | 80% (after 1 year) |
| 40°C | 85% (after 1 year) | 65% (after 1 year) |
| 60°C | 75% (after 1 year) | 60% (after 3 months) |
Table 3: Estimated recoverable capacity when storing Li-ion for one year at various temperatures
Elevated temperature hastens permanent capacity loss. Not all Li-ion systems behave the same.
Most Li-ions charge to 4.20V/cell, and every reduction in peak charge voltage of 0.10V/cell is said to double the cycle life. For example, a lithium-ion cell charged to 4.20V/cell typically delivers 300–500 cycles. If charged to only 4.10V/cell, the life can be prolonged to 600–1,000 cycles; 4.0V/cell should deliver 1,200–2,000 and 3.90V/cell should provide 2,400–4,000 cycles.
On the negative side, a lower peak charge voltage reduces the capacity the battery stores. As a simple guideline, every 70mV reduction in charge voltage lowers the overall capacity by 10 percent. Applying the peak charge voltage on a subsequent charge will restore the full capacity.
In terms of longevity, the optimal charge voltage is 3.92V/cell. Battery experts believe that this threshold eliminates all voltage-related stresses; going lower may not gain further benefits but induce other symptoms(See BU-808b: What causes Li-ion to die?) Table 4 summarizes the capacity as a function of charge levels. (All values are estimated; Energy Cells with higher voltage thresholds may deviate.)
| Charge Level* (V/cell) | Discharge Cycles | Available Stored Energy ** |
|---|---|---|
| [4.30] | [150–250] | [110–115%] |
| 4.25 | 200–350 | 105–110% |
| 4.20 | 300–500 | 100% |
| 4.13 | 400–700 | 90% |
| 4.06 | 600–1,000 | 81% |
| 4.00 | 850–1,500 | 73% |
| 3.92 | 1,200–2,000 | 65% |
| 3.85 | 2,400–4,000 | 60% |
Table 4: Discharge cycles and capacity as a function of charge voltage limit
Every 0.10V drop below 4.20V/cell doubles the cycle but holds less capacity. Raising the voltage above 4.20V/cell would shorten the life. The readings reflect regular Li-ion charging to 4.20V/cell.
Guideline: Every 70mV drop in charge voltage lowers the usable capacity by about 10%.
Note: Partial charging negates the benefit of Li-ion in terms of high specific energy.
* Similar life cycles apply for batteries with different voltage levels on full charge.
** Based on a new battery with 100% capacity when charged to the full voltage.
Experiment: Chalmers University of Technology, Sweden, reports that using a reduced charge level of 50% SOC increases the lifetime expectancy of the vehicle Li-ion battery by 44–130%.
Most chargers for mobile phones, laptops, tablets and digital cameras charge Li-ion to 4.20V/cell. This allows maximum capacity, because the consumer wants nothing less than optimal runtime. Industry, on the other hand, is more concerned about longevity and may choose lower voltage thresholds. Satellites and electric vehicles are such examples.
For safety reasons, many lithium-ions cannot exceed 4.20V/cell. (Some NMC are the exception.) While a higher voltage boosts capacity, exceeding the voltage shortens service life and compromises safety. Figure 5 demonstrates cycle count as a function of charge voltage. At 4.35V, the cycle count of a regular Li-ion is cut in half.

Figure 5: Effects on cycle life at elevated charge voltages [2]
Higher charge voltages boost capacity but lowers cycle life and compromises safety.
Besides selecting the best-suited voltage thresholds for a given application, a regular Li-ion should not remain at the high-voltage ceiling of 4.20V/cell for an extended time. The Li-ion charger turns off the charge current and the battery voltage reverts to a more natural level. This is like relaxing the muscles after a strenuous exercise(See BU-409: Charging Lithium-ion)
Figure 6 illustrates dynamic stress tests (DST) reflecting capacity loss when cycling Li-ion at various charge and discharge bandwidths. The largest capacity loss occurs when discharging a fully charged Li-ion to 25 percent SoC (black); the loss would be higher if fully discharged. Cycling between 85 and 25 percent (green) provides a longer service life than charging to 100 percent and discharging to 50 percent (dark blue). The smallest capacity loss is attained by charging Li-ion to 75 percent and discharging to 65 percent. This, however, does not fully utilize the battery. High voltages and exposure to elevated temperature is said to degrade the battery quicker than cycling under normal condition. (Nissan Leaf case)
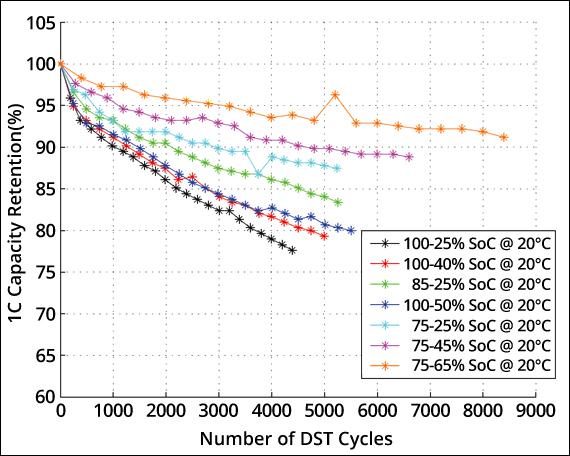
Figure 6: Capacity loss as a function of charge and discharge bandwidth* [3]
Charging and discharging Li-ion only partially prolongs battery life but reduces utilization.
- Case 1: 75–65% SoC offers longest cycle life but delivers only 90,000 energy units (EU). Utilizes 10% of battery.
- Case 2: 75–25% SoC has 3,000 cycles (to 90% capacity) and delivers 150,000 EU. Utilizes 50% of battery. (EV battery, new.)
- Case 3: 85–25% SoC has 2,000 cycles. Delivers 120,000 EU. Uses 60% of battery.
- Case 4: 100–25% SoC; long runtime with 75% use of battery. Has short life. (Mobile phone, drone, etc.)
* Discrepancies exist between Table 2 and Figure 6 on cycle count. No clear explanations are available other than assuming differences in battery quality and test methods. Variances between low-cost consumer and durable industrial grades may also play a role. Capacity retention will decline more rapidly at elevated temperatures than at 20ºC.
Only a full cycle provides the specified energy of a battery. With a modern Energy Cell, this is about 250Wh/kg, but the cycle life will be compromised. All being linear, the life-prolonging mid-range of 85-25 percent reduces the energy to 60 percent and this equates to moderating the specific energy density from 250Wh/kg to 150Wh/kg. Mobile phones are consumer goods that utilize the full energy of a battery. Industrial devices, such as the EV, typically limit the charge to 85% and discharge to 25%, or 60 percent energy usability, to prolong battery life(See Why Mobile Phone Batteries do not last as long as an EV Battery)
Increasing the cycle depth also raises the internal resistance of the Li-ion cell. Figure 7 illustrates a sharp rise at a cycle depth of 61 percent measured with the DC resistance method(See also BU-802a: How does Rising Internal Resistance affect Performance?) The resistance increase is permanent.

Figure 7: Sharp rise in internal resistance by increasing cycle depth of Li-ion [4]
Note: DC method delivers different internal resistance readings than with the AC method (green frame). For best results, use the DC method to calculate loading.
Figure 8 extrapolates the data from Figure 6 to expand the predicted cycle life of Li-ion by using an extrapolation program that assumes linear decay of battery capacity with progressive cycling. If this were true, then a Li-ion battery cycled within 75%–25% SoC (blue) would fade to 74% capacity after 14,000 cycles. If this battery were charged to 85% with same depth-of-discharge (green), the capacity would drop to 64% at 14,000 cycles, and with a 100% charge with same DoD (black), the capacity would drop to 48%. For unknown reasons, real-life expectancy tends to be lower than in simulated modeling(See BU-208: Cycling Performance)
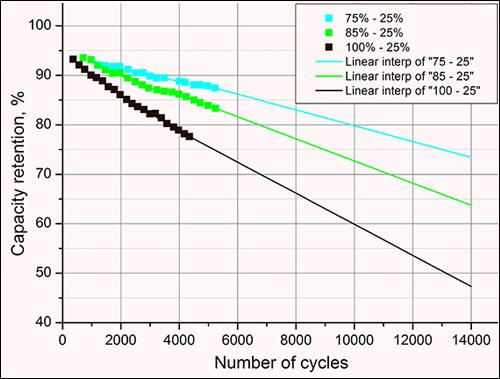
Figure 8: Predictive modeling of battery life by extrapolation [5]
Li-ion batteries are charged to three different SoC levels and the cycle life modelled. Limiting the charge range prolongs battery life but decreases energy delivered. This reflects in increased weight and higher initial cost.
Battery manufacturers often specify the cycle life of a battery with an 80 DoD. This is practical because batteries should retain some reserve before charge under normal use(See BU-501: Basics about Discharging, “What Constitutes a Discharge Cycle”) The cycle count on DST (dynamic stress test) differs with battery type, charge time, loading protocol and operating temperature. Lab tests often get numbers that are not attainable in the field.
What Can the User Do?
Environmental conditions, not cycling alone, govern the longevity of lithium-ion batteries. The worst situation is keeping a fully charged battery at elevated temperatures. Battery packs do not die suddenly, but the runtime gradually shortens as the capacity fades.
Lower charge voltages prolong battery life and electric vehicles and satellites take advantage of this. Similar provisions could also be made for consumer devices, but these are seldom offered; planned obsolescence takes care of this.
A laptop battery could be prolonged by lowering the charge voltage when connected to the AC grid. To make this feature user-friendly, a device should feature a “Long Life” mode that keeps the battery at 4.05V/cell and offers a SoC of about 80 percent. One hour before traveling, the user requests the “Full Capacity” mode to bring the charge to 4.20V/cell.
The question is asked, “Should I disconnect my laptop from the power grid when not in use?” Under normal circumstances this should not be necessary because charging stops when the Li-ion battery is full. A topping charge is only applied when the battery voltage drops to a certain level. Most users do not remove the AC power, and this practice is safe.
Modern laptops run cooler than older models and reported fires are fewer. Always keep the airflow unobstructed when running electric devices with air-cooling on a bed or pillow. A cool laptop extends battery life and safeguards the internal components. Energy Cells, which most consumer products have, should be charged at 1C or less. Avoid so-called ultra-fast chargers that claim to fully charge Li-ion in less than one hour.
References
[1] Courtesy of Cadex
[2] Source: Choi et al. (2002)
[3] B. Xu, A. Oudalov, A. Ulbig, G. Andersson and D. Kirschen, "Modeling of Lithium-Ion Battery Degradation for Cell Life Assessment," June 2016. [Online]. Available: https://www.researchgate.net/publication/303890624_Modeling_of_Lithium-Ion_Battery_Degradation_for_Cell_Life_Assessment.
[4] Source: Technische Universität München (TUM)
[5] With permission to use. Interpolation/extrapolation by OriginLab.
Last Updated: 11-Oct-2023

Batteries In A Portable World
The material on Battery University is based on the indispensable new 4th edition of "Batteries in a Portable World - A Handbook on Rechargeable Batteries for Non-Engineers" which is available for order through Amazon.com.
Find An Article
Table of Contents
-
Introduction
- BU-001: Sharing Battery Knowledge
- BU-002: Introduction
- BU-003: Dedication
-
Crash Course on Batteries
- BU-101: When Was the Battery Invented?
- BU-102: Early Innovators
- BU-103: Global Battery Markets
- BU-103a: Battery Breakthroughs: Myth or Fact?
- BU-104: Getting to Know the Battery
- BU-104a: Comparing the Battery with Other Power Sources
- BU-104b: Battery Building Blocks
- BU-104c: The Octagon Battery – What makes a Battery a Battery
- BU-105: Battery Definitions and what they mean
- BU-106: Advantages of Primary Batteries
- BU-106a: Choices of Primary Batteries
- BU-107: Comparison Table of Secondary Batteries
-
Battery Types
- BU-201: How does the Lead Acid Battery Work?
- BU-201a: Absorbent Glass Mat (AGM)
- BU-201b: Gel Lead Acid Battery
- BU-202: New Lead Acid Systems
- BU-203: Nickel-based Batteries
- BU-204: How do Lithium Batteries Work?
- BU-205: Types of Lithium-ion
- BU-206: Lithium-polymer: Substance or Hype?
- BU-208: Cycling Performance
- BU-209: How does a Supercapacitor Work?
- BU-210: How does the Fuel Cell Work?
- BU-210a: Why does Sodium-sulfur need to be heated
- BU-210b: How does the Flow Battery Work?
- BU-211: Alternate Battery Systems
- BU-212: Future Batteries
- BU-214: Summary Table of Lead-based Batteries
- BU-215: Summary Table of Nickel-based Batteries
- BU-216: Summary Table of Lithium-based Batteries
- BU-217: Summary Table of Alternate Batteries
- BU-218: Summary Table of Future Batteries
-
Packaging and Safety
- BU-301: A look at Old and New Battery Packaging
- BU-301a: Types of Battery Cells
- BU-302: Series and Parallel Battery Configurations
- BU-303: Confusion with Voltages
- BU-304: Why are Protection Circuits Needed?
- BU-304a: Safety Concerns with Li-ion
- BU-304b: Making Lithium-ion Safe
- BU-304c: Battery Safety in Public
- BU-305: Building a Lithium-ion Pack
- BU-306: What is the Function of the Separator?
- BU-307: How does Electrolyte Work?
- BU-308: Availability of Lithium
- BU-309: How does Graphite Work in Li-ion?
- BU-310: How does Cobalt Work in Li-ion?
- BU-311: Battery Raw Materials
-
Charge Methods
- BU-401: How do Battery Chargers Work?
- BU-401a: Fast and Ultra-fast Chargers
- BU-402: What Is C-rate?
- BU-403: Charging Lead Acid
- BU-404: What is Equalizing Charge?
- BU-405: Charging with a Power Supply
- BU-406: Battery as a Buffer
- BU-407: Charging Nickel-cadmium
- BU-408: Charging Nickel-metal-hydride
- BU-409: Charging Lithium-ion
- BU-409a: Why do Old Li-ion Batteries Take Long to Charge?
- BU-409b: Charging Lithium Iron Phosphate
- BU-410: Charging at High and Low Temperatures
- BU-411: Charging from a USB Port
- BU-412: Charging without Wires
- BU-413: Charging with Solar, Turbine
- BU-413a: How to Store Renewable Energy in a Battery
- BU-414: How do Charger Chips Work?
- BU-415: How to Charge and When to Charge?
-
Discharge Methods
- BU-501: Basics about Discharging
- BU-501a: Discharge Characteristics of Li-ion
- BU-502: Discharging at High and Low Temperatures
- BU-503: Determining Power Deliver by the Ragone Plot
- BU-504: How to Verify Sufficient Battery Capacity
-
"Smart" Battery
- BU-601: How does a Smart Battery Work?
- BU-602: How does a Battery Fuel Gauge Work?
- BU-603: How to Calibrate a “Smart” Battery
- BU-603a: Calibrating SMBus Batteries with Impedance Tracking
- BU-604: How to Process Data from a “Smart” Battery
- BU-605: Testing and Calibrating Smart Batteries
-
From Birth to Retirement
- BU-701: How to Prime Batteries
- BU-702: How to Store Batteries
- BU-703: Health Concerns with Batteries
- BU-704: How to Transport Batteries
- BU-704a: Shipping Lithium-based Batteries by Air
- BU-704b: CAUTION & Overpack Labels
- BU-704c: Class 9 Label
- BU-704d: NFPA 704 Rating
- BU-704e: Battery for Personal and Fleet Use
- BU-705: How to Recycle Batteries
- BU-705a: Battery Recycling as a Business
- BU-706: Summary of Do's and Don'ts
-
How To Prolong Battery Life
-
General
- BU-801: Setting Battery Performance Standards
- BU-801a: How to Rate Battery Runtime
- BU-801b: How to Define Battery Life
- BU-802: What Causes Capacity Loss?
- BU-802a: How does Rising Internal Resistance affect Performance?
- BU-802b: What does Elevated Self-discharge Do?
- BU-802c: How Low can a Battery be Discharged?
- BU-803: Can Batteries Be Restored?
- BU-803a: Cell Matching and Balancing
- BU-803b: What causes Cells to Short?
- BU-803c: Loss of Electrolyte
-
Lead Acid
- BU-804: How to Prolong Lead-acid Batteries
- BU-804a: Corrosion, Shedding and Internal Short
- BU-804b: Sulfation and How to Prevent it
- BU-804c: Acid Stratification and Surface Charge
- BU-805: Additives to Boost Flooded Lead Acid
- BU-806: Tracking Battery Capacity and Resistance as part of Aging
- BU-806a: How Heat and Loading affect Battery Life
-
Nickel-based
- BU-807: How to Restore Nickel-based Batteries
- BU-807a: Effect of Zapping
-
Lithium-ion
- BU-808: How to Prolong Lithium-based Batteries
- BU-808a: How to Awaken a Sleeping Li-ion
- BU-808b: What Causes Li-ion to Die?
- BU-808c: Coulombic and Energy Efficiency with the Battery
- BU-809: How to Maximize Runtime
- BU-810: What Everyone Should Know About Aftermarket Batteries
- BU-811: Assuring Minimum Operational Reserve Energy (MORE)
-
Battery Testing and Monitoring
- BU-901: Fundamentals in Battery Testing
- BU-901b: How to Measure the Remaining Useful Life of a Battery
- BU-902: How to Measure Internal Resistance
- BU-902a: How to Measure CCA
- BU-903: How to Measure State-of-charge
- BU-904: How to Measure Capacity
- BU-905: Testing Lead Acid Batteries
- BU-905a: Testing Starter Batteries in Vehicles
- BU-905b: Knowing when to Replace a Starter Battery
- BU-906: Testing Nickel-based Batteries
- BU-907: Testing Lithium-based Batteries
- BU-907a: Battery Rapid-test Methods
- BU-907b: Advancements in Battery Testing
- BU-907c: Cloud Analytics in Batteries
- BU-908: Battery Management System (BMS)
- BU-909: Battery Test Equipment
- BU-910: How to Repair a Battery Pack
- BU-911: How to Repair a Laptop Battery
- BU-915: Testing Battery with EIS
- BU-916: Deep Battery Diagnostics
- BU-917: In Search for Performance Transparency with Batteries
- BU-918: Battery Endurance Plan
- BU-919: Building a Matrix to test Batteries
- BU-920: Matrix Library
- BU-921: Testing Batteries by Multi-Model EIS
- BU-922: What Causes Starter Batteries to Fail?
- BU-923: Getting Deep Cycle Batteries Mission Ready
- BU-924: Battery Ecosystem
- BU-925: EIS Direct Drive, non-invasive Capacity Test
- BU-926: Battery Diagnostics, an Overlooked Requirement
-
Amazing Value of a Battery
- BU-1001: Batteries in Industries
- BU-1002: Electric Powertrain, then and now
- BU-1002a: Hybrid Electric Vehicles and the Battery
- BU-1002b: Environmental Benefit of the Electric Powertrain
- BU-1003: Electric Vehicle (EV)
- BU-1003a: Battery Aging in an Electric Vehicle (EV)
- BU-1004: Charging an Electric Vehicle
- BU-1005: Does the Fuel Cell-powered Vehicle have a Future?
- BU-1006: Cost of Mobile and Renewable Power
- BU-1007: Net Calorific Value
- BU-1008: Working towards Sustainability
- BU-1009: Battery Paradox - Afterword
-
Information
- BU-1101: Glossary
- BU-1102: Abbreviations
- BU-1103: Bibliography
- BU-1104: About the Author
- BU-1105: About Cadex (Sponsor)
- BU-1106: Author's Creed
- BU-1107: Disclaimer
- BU-1108: Copyright
-
Learning Tools
- BU-1501 Battery History
- BU-1502 Basics about Batteries
- BU-1504 Battery Test and Analyzing Devices
- BU-1505 Short History of Cadex
-
Battery Articles
- Perception of a Battery Tester
- Green Deal
- Risk Management in Batteries
- Predictive Test Methods for Starter Batteries
- Why Mobile Phone Batteries do not last as long as an EV Battery
- Battery Rapid-test Methods
- How to Charge Li-ion with a Parasitic Load
- Ultra-fast Charging
- Assuring Safety of Lithium-ion in the Workforce
- Diagnostic Battery Management
- Tweaking the Mobile Phone Battery
- Battery Test Methods
- Battery Testing and Safety
- How to Make Battery Performance Transparent
- Battery Diagnostics On-the-fly
- Making Battery State-of-health Transparent
- Batteries will eventually die, but when and how?
- Why does Pokémon Go rob so much Battery Power?
- How to Care for the Battery
- Tesla’s iPhone Moment — How the Powerwall will Change Global Energy Use
- Painting the Battery Green by giving it a Second Life
- Charging without Wires — A Solution or Laziness
- What everyone should know about Battery Chargers
- A Look at Cell Formats and how to Build a good Battery
- Battery Breakthroughs — Myth or Fact?
- Rapid-test Methods that No Longer Work
- Shipping Lithium-based Batteries by Air
- How to make Batteries more Reliable and Longer Lasting
- What causes Lithium-ion to die?
- Safety of Lithium-ion Batteries
- Recognizing Battery Capacity as the Missing Link
- Managing Batteries for Warehouse Logistics
- Caring for your Starter Battery
- Giving Batteries a Second Life
- How to Make Batteries in Medical Devices More Reliable
- Possible Solutions for the Battery Problem on the Boeing 787
- Impedance Spectroscopy Checks Battery Capacity in 15 Seconds
- How to Improve the Battery Fuel Gauge
- Examining Loading Characteristics on Primary and Secondary Batteries
-
Language Pool
- BU-001: Compartir conocimiento sobre baterías
- BU-002: Introducción
- BU-003: Dedicatoria
- BU-104: Conociendo la Batería
- BU-302: Configuraciones de Baterías en Serie y Paralelo
-
Batteries in a Portable World book
- Change-log of “Batteries in a Portable World,” 4th edition: Chapters 1 - 3
- Change-log of “Batteries in a Portable World,” 4th edition: Chapters 4 - 10