Rechargeable batteries play an important role in our lives and many daily chores would be unthinkable without the ability to recharge. The most common rechargeable batteries are lead acid, NiCd, NiMH and Li-ion. Here is a brief summary of their characteristics.
- Lead Acid – This is the oldest rechargeable battery system. Lead acid is rugged, forgiving if abused and is economically priced, but it has a low specific energy and limited cycle count. Lead acid is used for wheelchairs, golf cars, personnel carriers, emergency lighting and uninterruptible power supply (UPS). Lead is toxic and cannot be disposed in landfills.
- Nickel-cadmium – Mature and well understood, NiCd is used where long service life, high discharge current and extreme temperatures are required. NiCd is one of the most rugged and enduring batteries; it is the only chemistry that allows ultra-fast charging with minimal stress. Main applications are power tools, medical devices, aviation and UPS. Due to environmental concerns, NiCd is being replaced with other chemistries, but it retains its status in aircraft due to its good safety record.
- Nickel-metal-hydride – Serves as a replacement for NiCd as it has only mild toxic metals and provides higher specific energy. NiMH is used for medical instruments, hybrid cars and industrial applications. NiMH is also available in AA and AAA cells for consumer use.
- Lithium-ion – Li-ion is replacing many applications that were previously served by lead and nickel-based batteries. Due to safety concerns, Li-ion needs a protection circuit. It is more expensive than most other batteries, but high cycle count and low maintenance reduce the cost per cycle over many other chemistries.
Table 1 compares the characteristics of the four commonly used rechargeable battery systems, showing average performance ratings at time of publication. Li-ion is divided into different types, named by their active materials, which are cobalt, manganese, phosphate and titanate. (See BU-205: Types of Lithium-ion)
Missing from in the list is the popular lithium-ion-polymer that gets its name from the unique separator and electrolyte system. Most are a hybrid version that shares performance with other Li-ion. Also missing is the rechargeable lithium-metal, a battery that, once the safety issues are resolved, has the potential of becoming a battery choice with extraordinarily high specific energy and good specific power. The table only addresses portable batteries and excludes large systems that resemble a refinery.
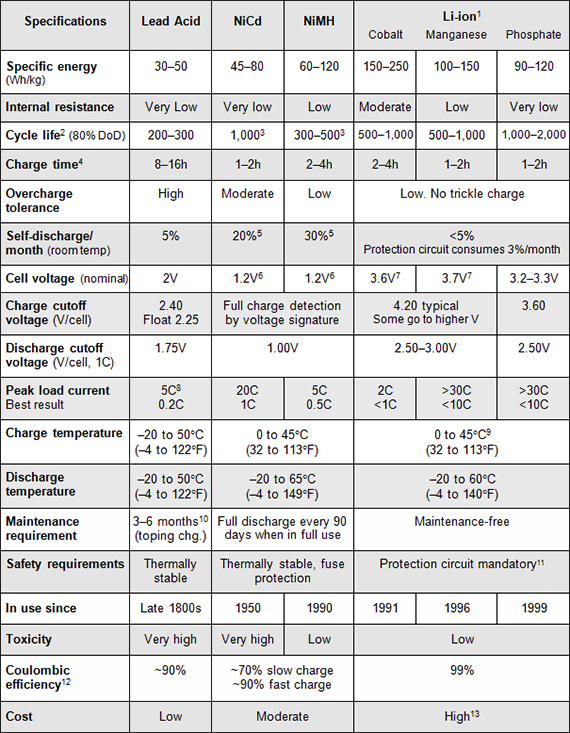
The figures are based on average ratings of commercial batteries at time of publication. Specialty batteries with above-average ratings are excluded.
- Combining cobalt, nickel, manganese and aluminum raises energy density up to 250Wh/kg.
- Cycle life is based on the depth of discharge (DoD). Shallow DoD prolongs cycle life.
- Cycle life is based on battery receiving regular maintenance to prevent memory.
- Ultra-fast charge batteries are made for a special pupose. (See BU-401a: Fast and Ultra-fast Chargers)
- Self-discharge is highest immediately after charge. NiCd loses 10% in the first 24 hours, then declines to 10% every 30 days. High temperature and age increase self-discharge.
- 1.25V is traditional; 1.20V is more common. (See BU-303: Confusion with Voltages)
- Manufacturers may rate voltage higher because of low internal resistance (marketing).
- Capable of high current pulses; needs time to recuperate.
- Do not charge Li-ion below freezing. (See BU-410: Charging at High and Low Temperatures)
- Maintenance may be in the form of equalizing or topping charge* to prevent sulfation.
- Protection circuit cuts off below about 2.20V and above 4.30V on most Li-ion; different voltage settings apply for lithium-iron-phosphate.
- Coulombic efficiency is higher with quicker charge (in part due to self-discharge error).
- Li-ion may have lower cost-per-cycle than lead acid.
* Topping charge is applied on a battery that is in service or storage to maintain full charge and to prevent sulfation on lead acid batteries.

Comments
Looking for comments from the previous website?
Comments from the previous website are not compatible with our new commenting system but we have preserved them so our users can still reference and make use the information in them.

All materials used in Li ion cell